PRODUCT OVERVIEW
We offer high-volume blood collection devices, kits, and sample tracking services that can be customized for a broad range of clinical trials, wellness, and clinical-grade blood sampling. Our HALO™ technology is bladeless, and a virtually painless way to collect whole, capillary blood that can be processed at any central lab.
HOW TO VIDEO
OUR TECHNOLOGY & PRODUCTS
Our virtually painless devices have always been high volume with the ability to collect up to 500 µL.

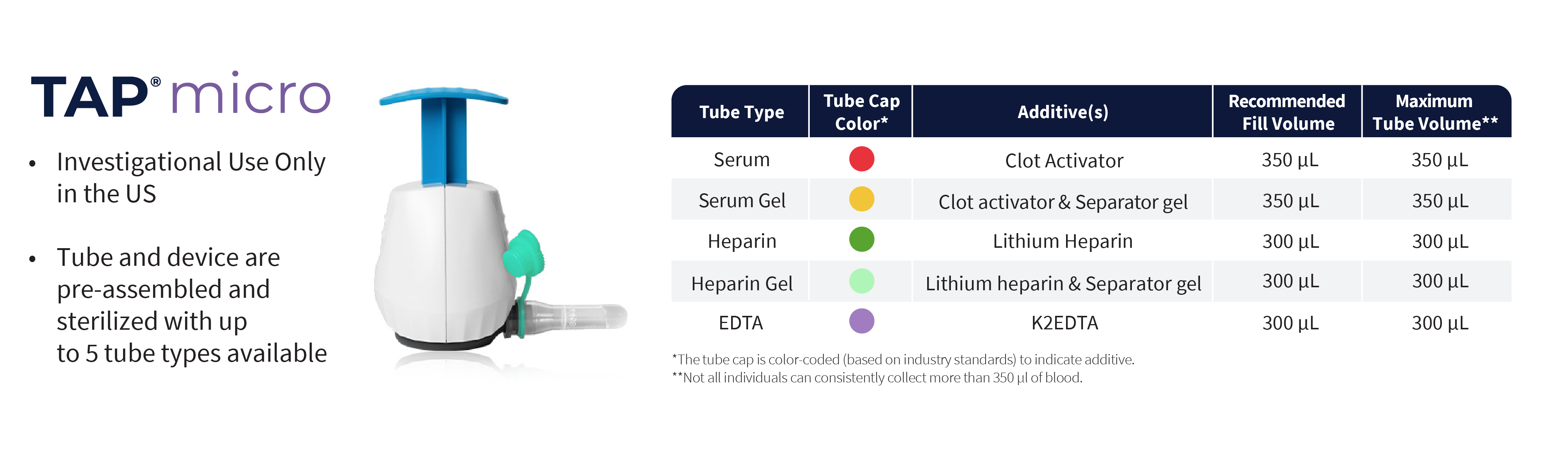
TAP® Micro
- CE Marked
- Investigational Use Only in the US
- Pre-attached collection tubes
- HALO™ Technology bladeless microneedle array

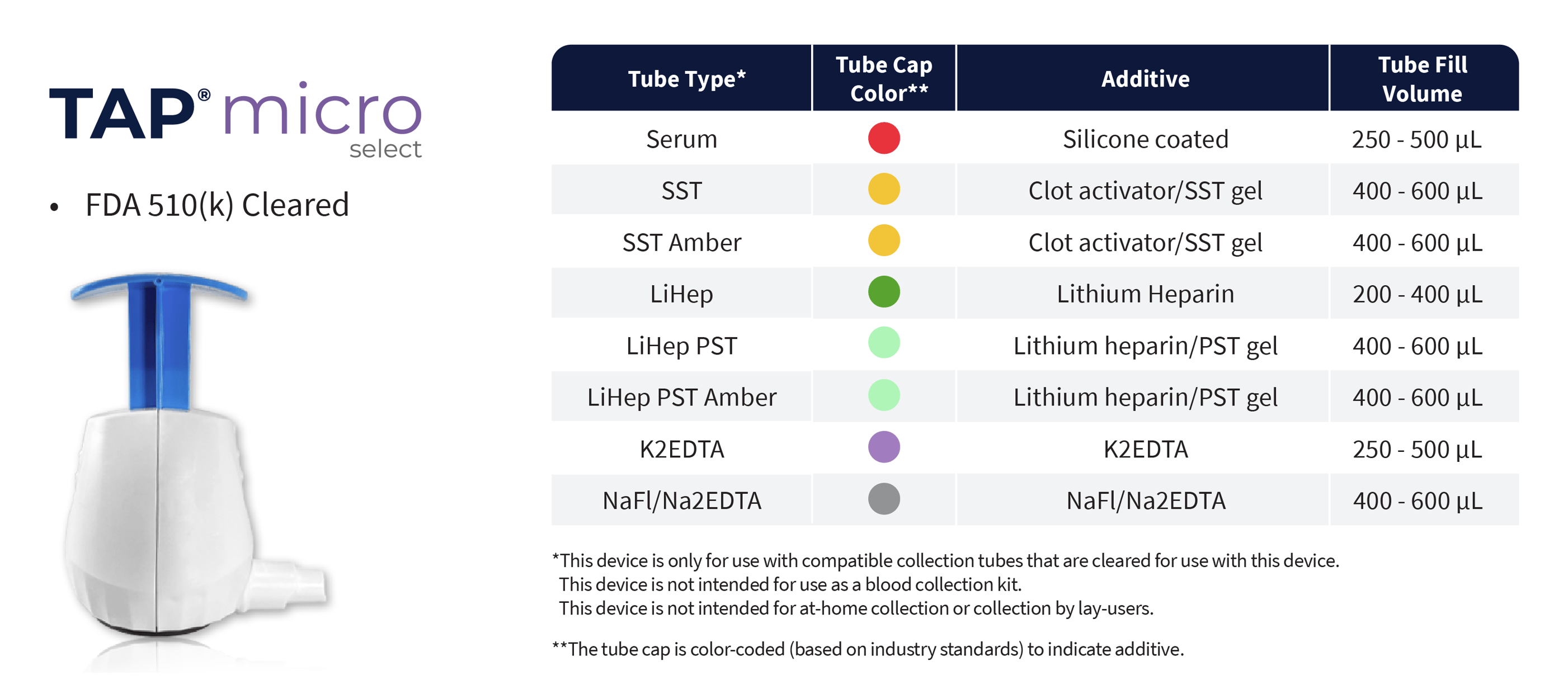
TAP® Micro Select
- FDA 510(k) Cleared
- CE Marked
- HALO™ Technology, bladeless microneedle array

HALO™ TECHNOLOGY
- Bladeless microneedle array
- Microneedles thinner than an eyelash
- Virtually painless compared to fingerstick, venipuncture, and competitive products
OUR COMMITMENT TO QUALITY
YourBio Health is committed to providing products that improve the quality, convenience, and cost-effectiveness of remote blood collection.
TAP® Micro Select is FDA 510(k) cleared and CE Marked.
TAP® Micro is CE Marked.
TAP® Micro is not FDA 510(k) cleared.
Manufactured in FDA Registered and ISO Certified facility.
ISO 13485